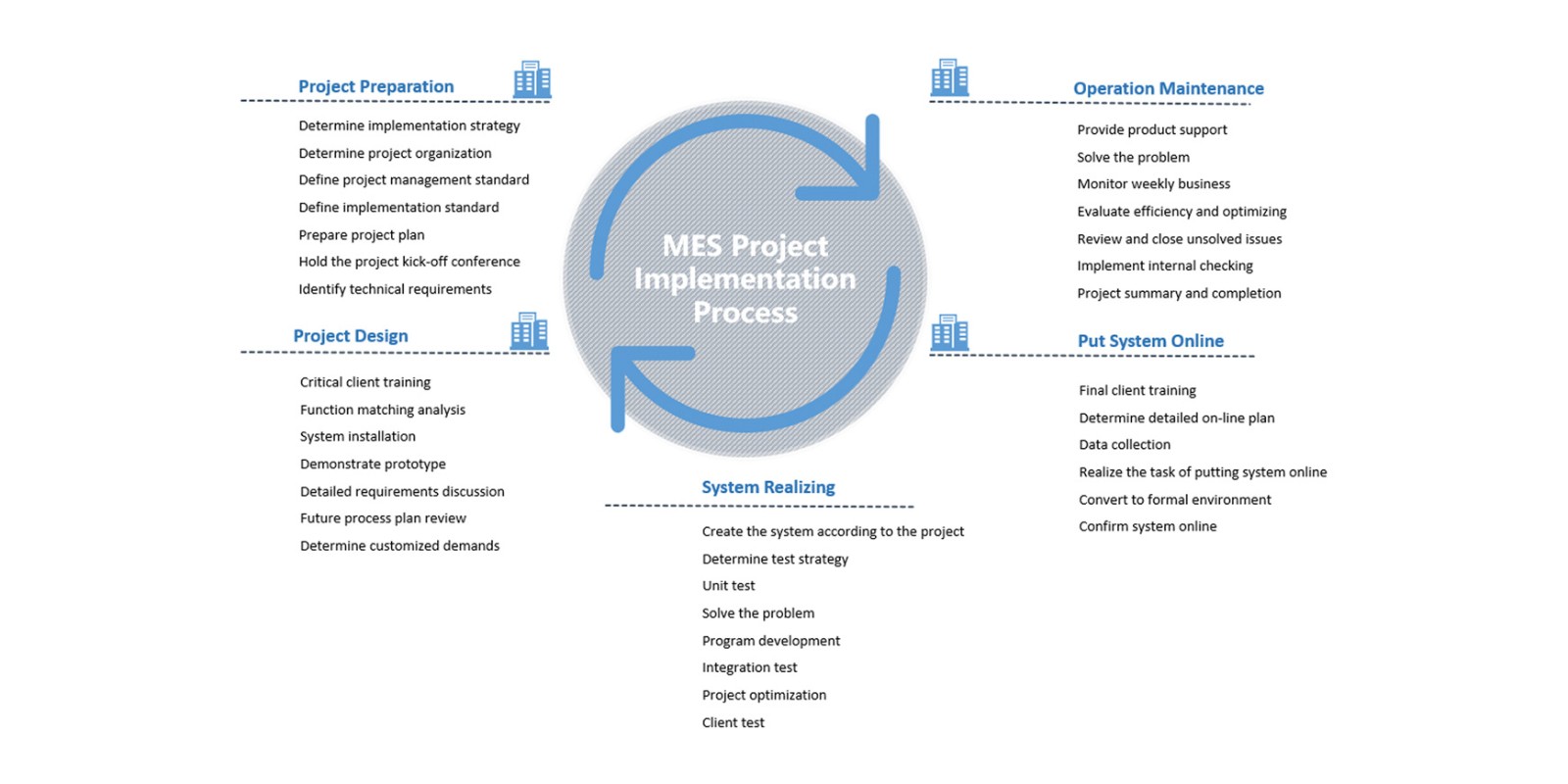
The Manufacturing Execution System (MES) is a workshop-level management software guiding and control of all the steps and operations in production process as well as collection and analysis of data generated during production process, used to improve production efficiency, compliance and stability. AUSTAR reached strategic cooperation with several leading MES suppliers, such as WERUM, ProLeiT, Rockwell and SIEMENS etc.
The MES system helps clients realize management control in the area of production order, production formula, accurate weighing, batch process, electronic work guiding, electronic batch records, product quality, workshop equipment and production kanban etc.

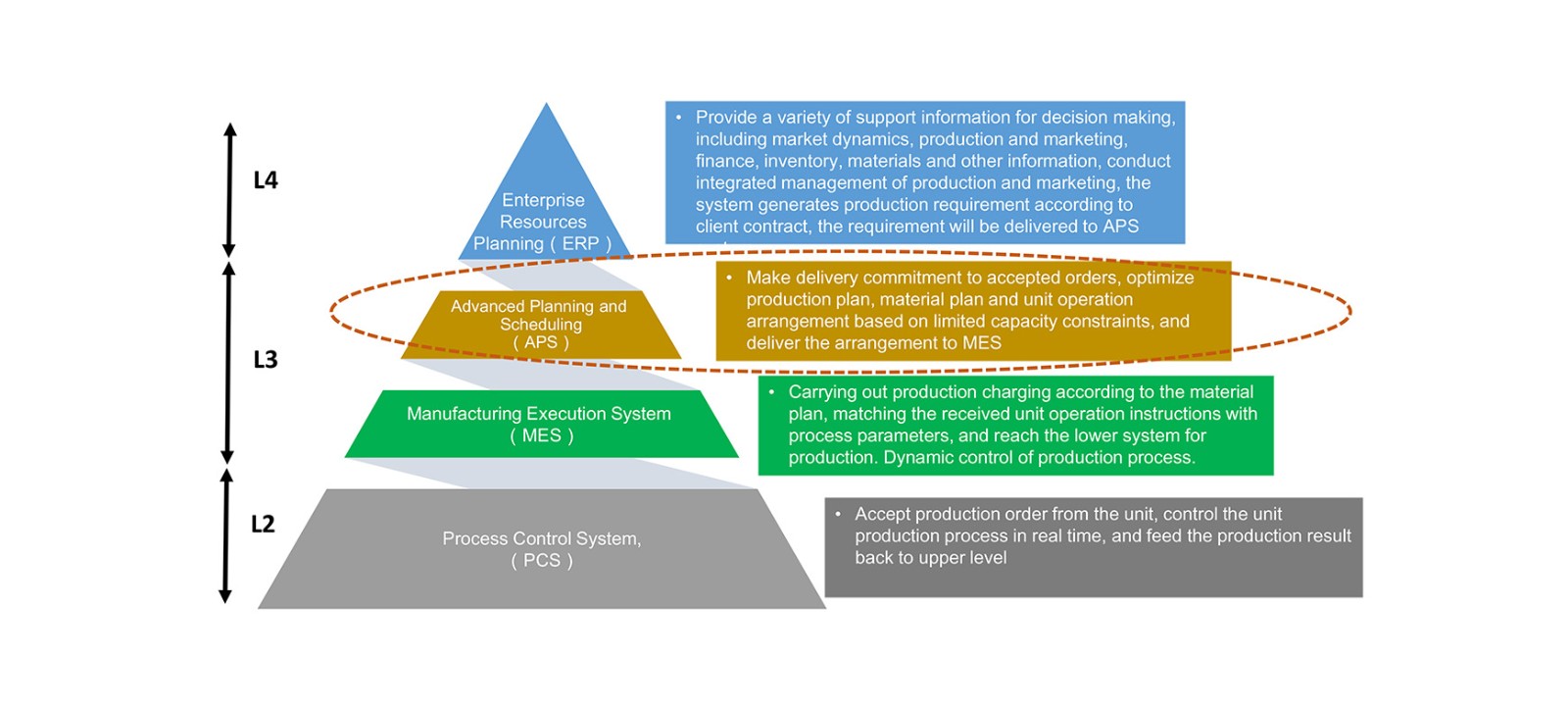
Advanced Planning and Scheduling (APS) is used to improve enterprise's factory-level scheduling capability by carrying out lean scheduling according to actual conditions of factory capacity and inventory with consideration of various production constraints.
The APS software has the functions of planning and scheduling, order capacity prediction and order progress follow-up etc. It firstly receives ERP order plan, inventory status and information of actual situation of workshop equipment, materials and personnel etc., then it conducts data integration, processing and decomposition and makes the optimized operation arrangement according to the specific algorithm with the arrangement result transmitted to manufacturing execution system for execution. APS helps enterprises achieve data circulation among planning, supply chain and production & manufacturing, making itself an indispensable part of the information architecture of enterprises.

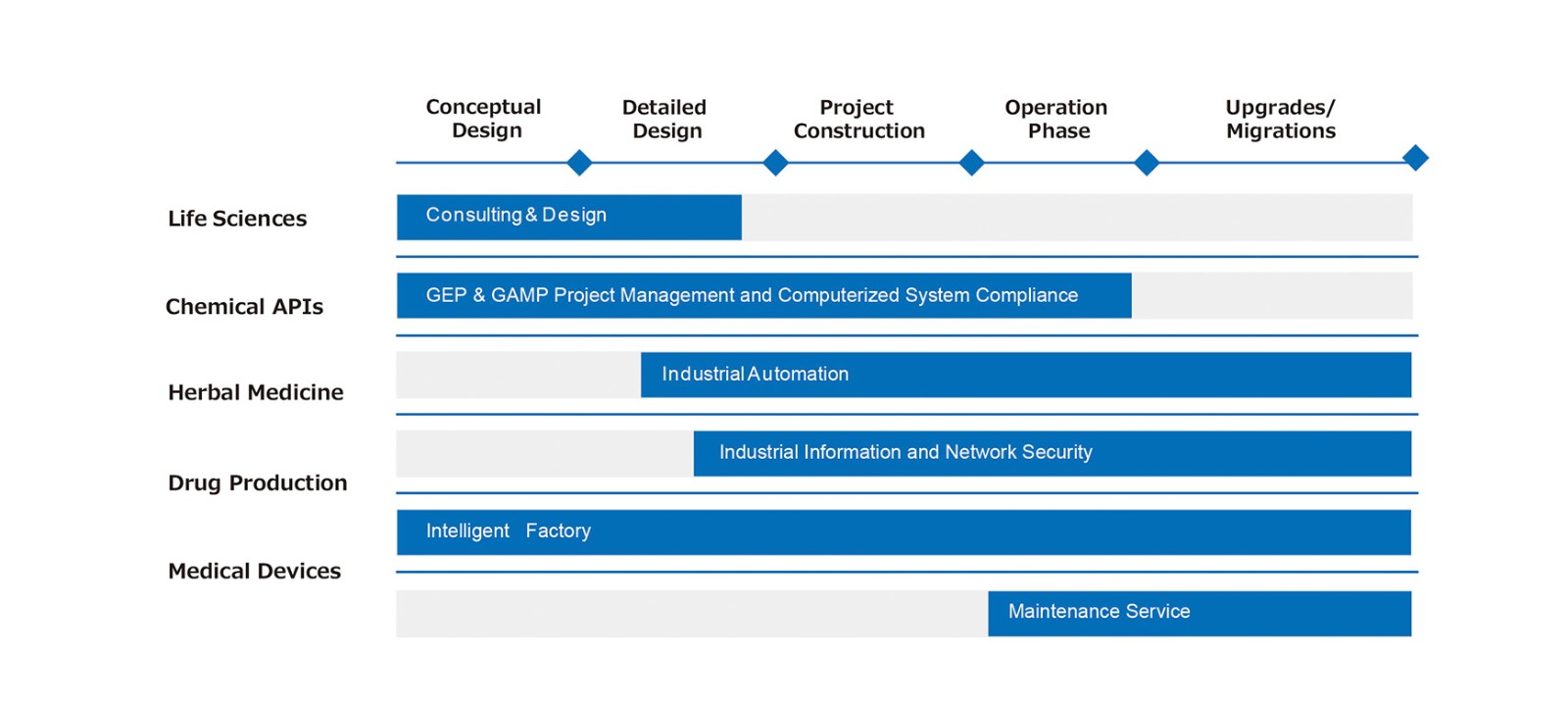
With its experienced team in industry process, automation, information and quality control, AUSTAR could help clients in the aspects of process optimization, engineering design optimization, project implementation and management and operation & maintenance. AUSTAR models and standardizes complex processes into a stable, visible and compliant automated manufacturing process which covers the main process and auxiliary process and is widely used in API, preparations, biopharmaceutical, traditional Chinese medicine and other drugs. Through precise and optimized control scheme to ensure product quality, improve production efficiency, reduce production cost and guarantee production safety.

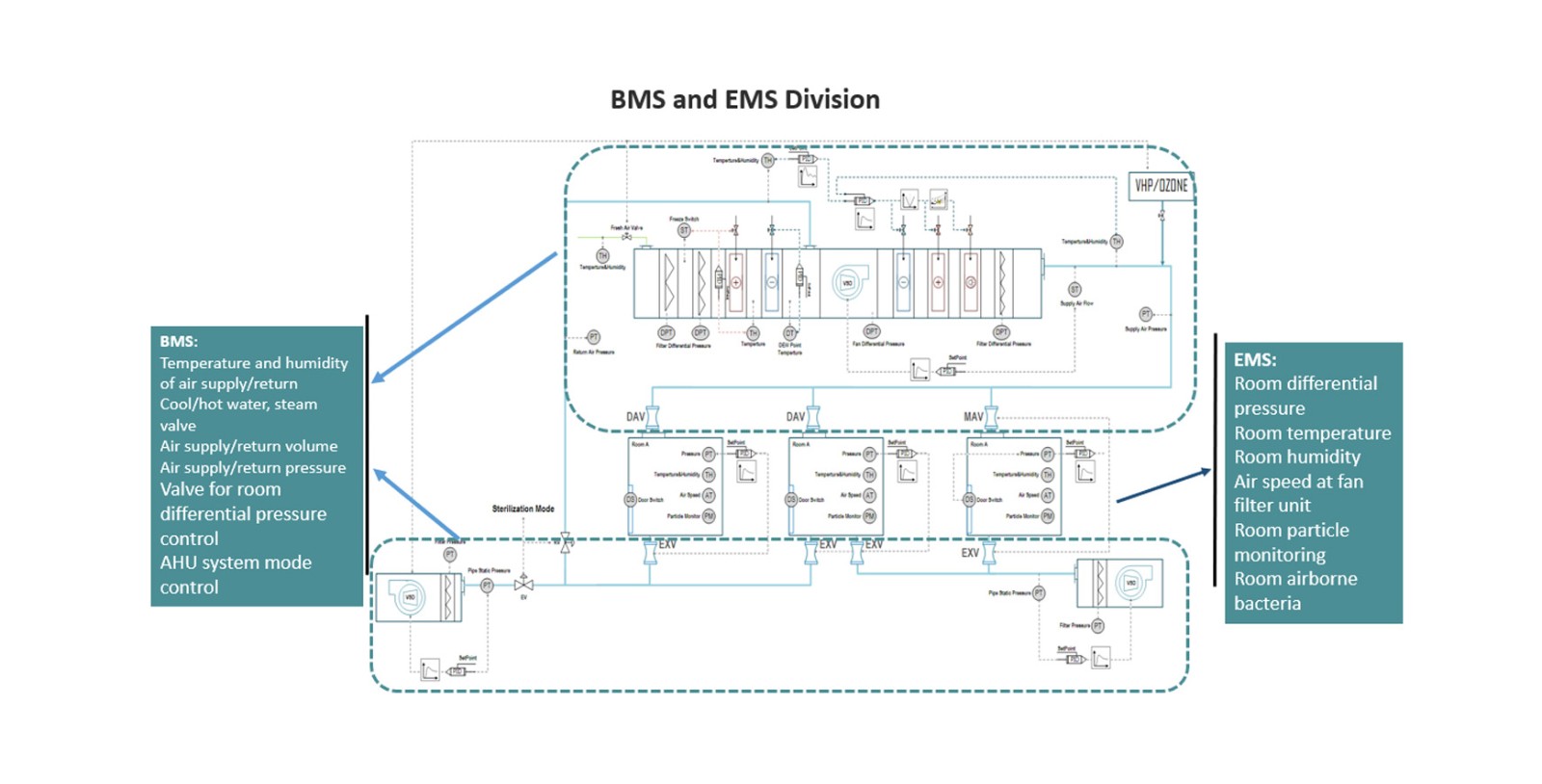
Automation solutions of utilities includes building management system (BMS), GMP critical parameters monitoring system (GMS), public circulating water system, industrial steam, air pressure system, weak current system integration and equipment system upgrade, etc. Advanced perfect controlling systems assists to reduce energy consumption, guarantee clean room environment, and reduce equipment failure and maintenance & operation cost.
The automation solutions for utilities developed by AUSTAR are widely used in monoclonal antibody biopharmaceutical enterprises, vaccine biopharmaceutical enterprises and aseptic preparations. As a integrated solution provider, AUSTAR utility engineering automation provides full life cycle services covering consulting, design, engineering implementation, operation and maintenance. The building management system provides HVACR all-round optimization control and energy saving management. With years of experiences in project practice, AUSTAR realizes high-precision and stability temperature, humidity and pressure difference control of AHU END of HVAC and high-efficiency operation of multi-system coupling group control. The GMP data monitoring system, based on the risk analysis method of comprehensive data compliance, provides data management for collection, recording, archiving and inquiry of enterprise environmental parameters, clean engineering parameters, personnel access information and process parameters.
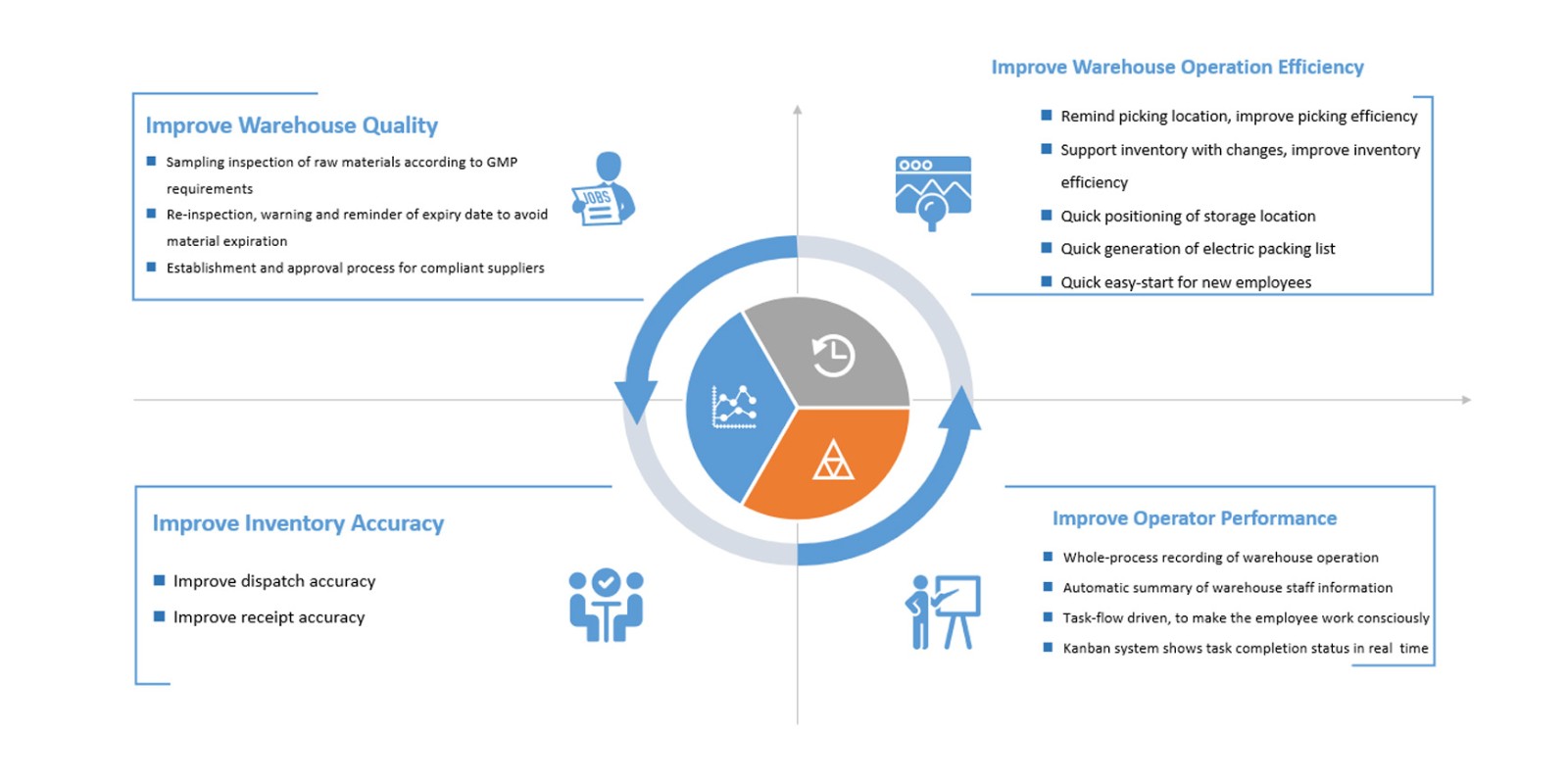
The Warehouse Management System (WMS) manages in-and-out material warehousing business including warehousing of procurement, warehousing of production, warehousing of production, warehousing of sales and other warehousing modes. The system will arrange the material out of the warehouse automatically according to the principle of "priority for scattered materials, first in and first out and first out of materials which are close to expiration date" to prevent occurrence of sluggish material. The system manages material batch status, quality inspection and material release, including raw materials, packaging materials, in-process products and finished products, accomplishing on-line management of sampling, inspection and release of in-process products and finished products.
AU-WMS warehouse management system is independently developed by AUSTAR. With reference to relevant regulations of domestic GMP, EU GMP and FDA and years of experience in verification and consultation, AUSTAR constantly optimizes the system according to clients' needs, and finally forms this professional warehouse management system suitable for pharmaceutical enterprises. AU-WMS provides standardized interface to solve the interaction of information between various platforms and systems with quick response.





 Search
Search 中文
中文






